One of the most important questions that every scientist learns to ask is “How do you know that…?” As scientists, we are trained to be skeptical. When we consider a bit of research done by a colleague, before we are inclined to believe the data, we need to be sure that they conducted the right experiments and that those experiments were done correctly. This doesn’t mean that scientists are stubborn or closed-minded. The reality is quite the opposite. Scientists are ready to incorporate new ideas and new results but first we need to know that the data are real. That’s what being a skeptic is all about: reserving judgment until you know all the facts.
The question “How do you know that..?” is one of the intellectual tools we use when considering whether or not data are real or not. This question has two parts: 1) how do you measure the thing that you interested in and 2) how do you know the effect you are seeing is actually based on what you think it is? What type of comparisons do you need to make in order to test the effect you’re interested in?
The first point of the question relies on special tools, equipment/technology, and experimental setups that are used to take measurements. For example, if you want to know how much a mouse likes taking a drug, then you need a way to measure how much drug it takes and how often it takes the drug (more on this in a bit). Today, I’ll go over a few of the tools that we use in addiction research.
The second part is more important (and more difficult to explain) but is really at the heart of the scientific method. It is all about experimental design and making sure you make the proper comparisons and analyses. I won’t discuss these details any more right now but will save this discussion for a future post.
Instead, let’s take a look at a few of the tools a scientist studying drug addiction has in his/her toolbox.
Locomotor Activity Test
The psychostimulants amphetamine and cocaine act in very similar ways and have very similar effects on the brain. We know that stimulants sort of “amp you up” or make you feel like you have more energy. Think of how you feel after drinking too much coffee. And what do you do when you have more energy? You tend to move around more (maybe you feel a little twitchy/antsy after too much of that coffee…). The same thing happens to mice and rats.

Locomotor Activity Test Chamber with a mouse. Image from UC-Davis Mind Institute (http://www.ucdmc.ucdavis.edu/mindinstitute/).
We can measure the amount of movement using a locomotor activity test. This test uses a special piece of equipment that uses light beams and a light-sensitive detector. Whenever the animal moves around the test box, the light beams are broken and the detector records that information. One way to analyze the data is by simply plotting beam-breaks (photo-cell counts are the same thing) that occur over the time of the test period. This way you have a measure of how much the animal moves around in a certain amount of time (more beam-breaks/time unit equals greater movement). A more sophisticated analysis of this same data can actually give you information on where in the box the animal spends its time. Does is just pace back and forth in a small area of the box or does it explore the entire chamber? This type of exploratory behavior data is valuable information and can be useful to other fields that may or may not study drug addiction. The general test for this exploratory behavioral analysis, regardless of speed of the movement caused by drugs, is the open field test.

Multiple test boxes with a computer that collects the data. Image from Douglas Mental Health Institute (http://www.douglas.qc.ca/page/neurophenotyping-motor-function).
An interesting phenomenon has been identified with psychostimulants. If you give an animal an injection of cocaine it will move around more compared to regular animals. But if you give it another dose of cocaine the next day it will move around even more than it did on the first day. This is called locomotor sensitization and is an important property of psychostimulants like amphetamine and cocaine.
The graphs below are real data that I took from a figure from one of our lab’s papers so you can see what locomotor sensitization looks like.
It’s a little hard to read but there are two groups of animals: one that receives cocaine injections (the top line) and the other that receives saline injections (the bottom line). Saline is a saltwater solution that is a standard control solution that has no biological effects. Each data point represents an average of several animals from each group. The baseline graph shows the locomotor activity before injections (no differences). As you can see, at day 1 the cocaine animals are already moving more than the saline group. This increase in movement continues over the 14 days of the experiment, evidence of locomotor sensitization.
This video shows an analysis of locomotor activity using video tracking software instead of light-beam breaks.
Self-Administration
Locomotor activity is all good and well but not all drugs of abuse cause locomotor sensitization. More directly related to addiction in humans, how do we even know if the animal likes the drug or wants to take the drug? Humans addicts crave the drug and compulsively use it, meaning the desire to do the of the drug overpowers the addict’s self-control. Is there a way we can study this type of drug-taking behavior in animals? The answer is yes!
Self-administration is a very versatile and powerful technique used throughout the addiction field. This technique allows the animal to control whenever it takes the drug and however much it wants. We can study many different aspect of drug taking using self-administration.

A diagram for a typical self-administration chamber. Image from Med Associates (http://www.med-associates.com/).
The basic idea is is simple: The rodent (mouse or rat) is placed in a chamber and presented with two levers. If the mouse the presses one lever (the active lever) it receives a dose of drug but if it presses the other lever (inactive lever) it does not. The self-administration sessions are run for a set period of time and the number of presses is recorded for each lever. Over the course of several days the animal steadily increases the amount of lever presses, thus the amount of drug it takes. Meaning the animal learns how to take drug and then takes more and more of it. Just like a human addict would do!
Alternatively, the mouse can poke its nose at a special hole that acts just like the active lever. I’ll use “lever press” and “nose poke” interchangeably because they essentially mean the same thing.
Here’s a little cartoon I found on YouTube of a rat that is self-administering nicotine.
Here’s another video that shows a real mouse self-administering a natural reward (meaning not a drug of abuse but food in this case).
There are several important variations to this basic idea that help scientists to not only make the experiments easier to control and data better/easier to analyze, but allow different aspects of drug taking to be studied.
For example if you are studying alcohol addiction, then when the mouse presses the lever a spout may appear that allows the animal to drink the alcohol (the inactive lever produces a bottle of water only). This is perfect for testing alcohol self-administration because both humans and mice drink alcohol. But what if you want to study heroin or cocaine self-administration? Humans (nor mice) drink or eat these drugs. So how does the drug get delivered to the mouse when it presses the lever?
The answer is intravenous self-administration. In this version, a small surgery is performed where a small tube (a cathether) is threaded into the jugular vein of the animal. This tube is fixed to the mouse back and attached to another tube that is part of the self-administration apparatus. This time when the mouse hits the lever, a dose of drug is pumped directly into its vein! See the diagram and videos above for more details.
Intravenous self-administration has several advantages.
- As explained above, it allows us to deliver drugs to animals that won’t take them orally.
- It allows the drug to act immediately on the animal because the drug is being delivered directly into the bloodstream.
- It allows us to control the dose of the drug. When the mouse hits the lever (or nose pokes) it receives a fixed amount of drug that the scientist decides on ahead of time. That way we know how much total drug the mouse takes during a single self-administration session.
- There is no variability in whether the animal is receiving the full dose or not. For example, if the lever press results in a food pellet, there is no guarantee the animal will eat the whole thing. But if you set the self-administration apparatus to deliver 0.5mg of heroin every time the lever is pressed, then there is no doubt if the full 0.5mg dose is delivered to mouse ever time.
Warning: not for the squeamish! This video shows you how to do the catheter implantation surgery on a mouse that will be used for intravenous self-administration!
Finally, best of all, self-ad can be used to address many different types of questions related to different stages in the addiction cycle. Here I briefly describe some of the more common experimental questions and applications that self-ad can help to address.
- Initial use and escalation of use. How much will the animal take when it is first exposed to the drug? Will the animal reach a ceiling in the amount of drug it will take in a single session?
- Maintenance of drug taking. One cool variation is you can make it more difficult for the animal to get the same dose of drug. This is called a progressive ratio self-administration. For example, the animal may need to press the lever 5 times before it receives a dose. You can keep increasing the number of presses during each session to see how hard the animal will work for a dose. One way this experiment can be interpreted is how badly does the animal want the drug? Some animals will press the lever many, many times just to get a small dose. This type of behavior is similar to the intense cravings that human addicts can experience.
- Extinction and Relapse. You can run a special type of experiment where you run a self-administration experiment like normal and then change it so that the active lever no longer gives the animal a dose of drug. Eventually, the animal presses the lever less and less as it learns that it will no longer get the drug. This is called extinction of self-administration. This is like being in a rehab clinic where you are prevented from taking the drug. However, after the extinction sessions, if the scientist gives the animal another does of drug this will causes animal to start pressing the lever at high rates again. This a called reinstatement of self-administration and is model of relapse. What other types of conditions or factors can cause reinstatement (relapse behavior)? This situation is just like an abstinent cocaine addict who may not be craving cocaine but if he/she takes even a single hit, this can be sufficient for that person to sink back into full-blown addiction.
Let’s take a look at some real data. The graph below is from a paper from our group that looks at oxycodone self-administration in mice.

Oxycodone self-administration by adult and adolescent mice. (Zhang Y et al. Neuropsychopharmacol. 2009.)
This study is interested in comparing oxycodone self-administration between adult mice and adolescent mice. As you can see, the number of nose pokes at the active hole (remember, same thing as a lever presses) increases during the course of the experiment (don’t worry about FR1 vs FR3) while the inactive hole is ignored, because it does not result in drug administration. Note that the nose pokes are plotted over the time of the administration sessions (2 hours) and that 9 sessions are run (one every day).
Microdialysis
The types of experiments I’ve described so far are great ways of studies addictive behaviors but they don’t really tell you about what’s going on in the brain. These behavior experiments are useful in themselves but they are much more powerful if they can be combined with another type of experiment that gives you a window into what’s changing in the brain at the same time as the behaviors.
In my post Synapse to it, I described how neurotransmitters are released by the pre-synaptic neurons into the synaptic cleft so that they can act on receptors located on the post-synaptic neuron. Using microdialysis, you can sample the fluid that exists in the synaptic cleft and actually measure the amount of neurotransmitters being released!
This is an extremely difficult and very technically complicated technique and I will only go into the basics about it. First, the microdialysis probe is surgically placed into a region of the brain that you are interested in studying.
The microdialysis probe itself is like a very thin piece of tubing that allows the experimenter to flow fluid into it one side(inlet) and collect the fluid that flows out of the other side (outlet). At the tip of the probe (the part that’s actually inside the brain) is a special type of material that allows fluid from inside the brain to flow into the tubing (a semi-permeable membrane).
After the surgery, you run your behavioral experiment, and while you are doing that you start flowing fluid into the brain. The fluid that the microdialysis probe flows in is of a similar consistency to the fluid that exists naturally in the brain. As the fluid inside the probe moves through the tubing, it causes fluids in the brain to enter into the probe and through the tubing where it can be collected when it flows out of the tubing.
Let’s say you give an animal a drug that causes a neurotransmitter to be released in the brain region you are interested in. Then some of those released neurotransmitters will enter the microdialysis probe because some of the fluid that enters the probe is from the synaptic cleft.
You keep collecting fluid at different time points during your experiment. When the experiment is over, then you can use chemistry to determine what neurotransmitters are in the fluid you collected. Best of all, you can determine how much of those neurotransmitters you have! How you do actually use chemistry to do this is a very technical part of the procedure and is not important to this discussion.
And all that work gives you a nice graph of the neurotransmitters that are released at different times during your experiment.
Now for some real data. Below are figures from a paper that our lab produced that uses microdialysis to study release of the neurotransmitter dopamine.
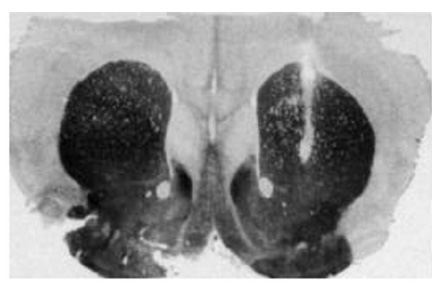
In this study, the effect of cocaine on dopamine release in a region of the brain called the caudate putamen is being studied. The first image shows you that the microdialysis probe was placed in the right area of the brain (the white line that pierces through the dark area is the tract in the caudate putamen). The graph shows that injection of cocaine (the arrows) causes an increase in dopamine release in this brain region. Interestingly, the dopamine levels have returned to normal by the end of the experiment. Note: C57Bl/6J is the strain of mouse used in this study.
These are just three of the techniques that are used in addiction research. But we scientists have very big toolboxes! I’ll to explain some more in a later post.
Feel free to contact me or comment if you have questions!
Thanks for reading 🙂





Interesting and informative,great work son!
It is also nice to have your music to listen to while reading.
LikeLiked by 1 person
Pingback: The Science of Stress and Addiction: A Mini-review of the Research, Part 1 – Dr. Simon Says Science
Pingback: Stress and Addiction Part 3: Molecular Changes – Dr. Simon Says Science
Pingback: The Scientist’s Toolbox: Techniques in Addiction Research, Part 2 – Dr. Simon Says Science
Pingback: Morphine and Oxycodone Affect the Brain Differently – Dr. Simon Says Science