 This is part three on my series of posts looking at Stress and Addiction. To recap: we’ve seen that, in laboratory studies, stress increases susceptibility to drug addiction. Stress not only increases the self-administration of drugs in adult rats but stress during an early age can have a long-lasting effect on drug-taking behavior. Today, we’ll wrap up by looking at some molecular changes that might help to explain why this effect exists. I’ll conclude by addressing some questions that might have occurred during the course of this discussion.
This is part three on my series of posts looking at Stress and Addiction. To recap: we’ve seen that, in laboratory studies, stress increases susceptibility to drug addiction. Stress not only increases the self-administration of drugs in adult rats but stress during an early age can have a long-lasting effect on drug-taking behavior. Today, we’ll wrap up by looking at some molecular changes that might help to explain why this effect exists. I’ll conclude by addressing some questions that might have occurred during the course of this discussion.
Paper #1 
The first paper is examining the effects of stress and cocaine on dopamine. Dopamine is a very important neurotransmitter. All drugs of abuse cause increases in dopamine in an important region of the brain called the mesolimbic pathway. I will discuss this system in detail in the next post but for now don’t worry about the details. All you need to know is that drugs can increase dopamine.
Dopamine levels can be measured directly in the brain using the technique microdialysis (I discuss this technique in more detail in my post The Scientist’s Toolbox: Techniques in Addiction). In this first paper, the scientists use a type of stress called foot-shock stress. It is very similar to tail-pinch stress (see Part 1). The animals are placed on a grid that is connected to an electrical supply. The scientists administer a small amount of electric current to the grid, which gives the animals feet a little shock and stresses them out.
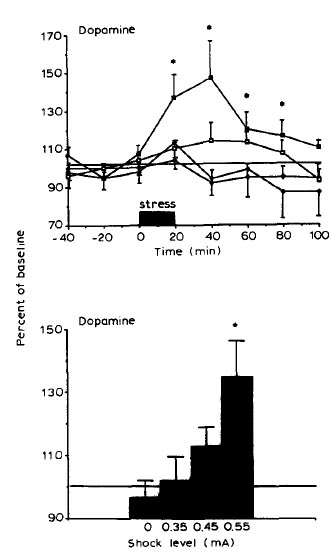
Figure 1.
The microdialysis technique was used on rats that underwent foot shock stress in order to measure dopamine levels (in a region of the brain called the striatum) after the stress test. As you can see in the top graph of Figure 1, foot shock stress causes an immediate increase in the amount of dopamine released and this eventually returns to normal. The different symbols mean different stress intensity with the most intense stress represented as black squares. Interestingly, as you can see in the lower graph of Figure 1, a more intense foot shock (ie a more intense stress) causes more dopamine to be released.
Remember that I said that cocaine also causes dopamine release? So maybe stress makes cocaine feel better because it works together with cocaine to create a larger release in dopamine than cocaine would by itself. Next, the investigators decided to test this idea.

Figure 2.
In this experiment, mice were exposed to a weak foot-shock stress then given an injection of cocaine and the amount of dopamine released was measured. Figure 2 shows much more dopamine was released in the striatum in rats that received cocaine + stress (black squares) compared to cocaine + no stress (white squares) or just stress by itself (black circles). Perhaps the hypothesis that stress makes cocaine more pleasurable because its boosts dopamine released might be true?
Paper #2

Recall from Part 1, that stress activates the HPA axis, which results in release of the stress hormone cortisol (corticosterone in rats and mice). But do drugs of abuse also activate the HPA axis? This next paper—done in lab that I work in—takes a look at this question.
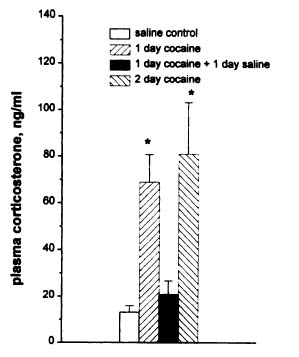
Figure 1.
Cocaine was given to rats under a number of different conditions. In the first experiment, cocaine effects on the HPA axis were examined in the short term (acute cocaine use). Rats were injected with either saline for two days, cocaine for 1 day, cocaine for 1 day and saline for 1 day, or cocaine for 2 days. After the injections, blood was drawn from the animals and the corticosterone in the serum was measured.
*Technical notes: 1) Serum is the liquid part of blood and it does not contain the red blood cells and clotting proteins. Serum is often used when measuring hormones in the blood. 2) Corticosterone can be measured multiple ways but this experiment used something called a radioimmunoassay (RIA). I’ll save the explanation of it for a future Scientist’s Toolbox post.
As you can see in Figure 1, immediately after the rats receive cocaine (either 1 day or 2 days) corticosterone increases. This means that cocaine has resulted in activation of the HPA axis. Interestingly, the animals received 1 day of cocaine and 1 day of saline did not show high corticosterone levels which means that the levels have returned to normal after the cocaine.
But what happens with repeated cocaine use (chronic cocaine use)? Addiction develops because of chronic use of the drug so are any changes occurring after many days of cocaine use?

Figure 2.
Interestingly, in Figure 2, corticosterone is high after 3 days of cocaine but after 14 days of cocaine corticosterone levels are much lower! What’s going on here? What these data suggest is that 14 days of cocaine use has caused a change in the HPA axis activity. The cocaine has activated the HPA axis so frequently the axis has compensated for this over activation. The activity of the HPA axis response has been blunted because of the repeated cocaine use.
This one small example of how drugs can cause long lasting molecular adaptations and changes in the brain. Perhaps this is why stress helps to make someone more vulnerable to addiction, because changes occur both at the level of dopamine release (paper #1) and in HPA axis activity (paper #2). Both drugs and stress have similar molecular effects that may work together! I’d like to very briefly discuss one more paper that combines both of these concepts.
Paper #3

This paper is complicated but I’m just going to present a small amount of the data. The key points you need to know is that the scientists are using social stress (see Part 1) in this paper for two key experiments 1) self-administration to measure cocaine taking behavior and 2) microdialysis to measure dopamine release. However, they also inject a chemical compound directly into the rat’s brain that blocks HPA axis activity. This chemical acts at the starting point in the HPA axis: the activity of CRF is blocked (the chemical name is abbreviated as CP). Let’s see what happens in this experiment!
*Technical notes: 1) the drug actually prevents the action of CRF interacting with its receptor. Chemicals that do this are called antagonists. Therefore, the scientists are injecting a CRF Receptor antagonist into the rat brains. 2) as a control, an inactive solution is also injected into some animals. This is called artificial cerebral spinal fluid (aCSF). For the drug studies, the correct comparision is CP vs aCSF.

Figure 1.
Like we saw in other papers, stress increases self-administration (Figure 1, black circles) compared to no stress (white triangles). However, when you give the CP at a high dose (light grey circles) compared to a low dose (dark grey circles) it reduces the self-administration! This means that blocking HPA axis activity reduces the effects of the stress on the cocaine self-administration. Cool!

Figure 2.
Next, they did a very similar experiment but only this time measure the interaction between stress, cocaine, and the CRF antagonist on dopamine release. The results are presented in Figure 2. Animals that were stressed and than given a dose of cocaine but not the CP (stress + cocaine + aCSF, black circles) released a large amount of dopamine compared to animals that were only given the cocaine injection (white triangles), which is consistent with findings from Paper #1. Amazingly animals that were stressed and then given cocaine + the anti-HPA axis drug CP showed reduced amounts of dopamine released at bot a low dose of CP (dark grey circles) and high dose (light grey circles). These experiments show that the effect of stress on cocaine taking behavior might be because the stress activates the HPA axis which causes more dopamine to be released.
*Technical note: I described this experiments very briefly but they are extremely technically challenging and probably required months of hard work just to make the two little graphs!
Summary
Finally, if we summarize the papers from Part 1, 2 and 3 we can come up with a little mechanism to help explain the different results from the different papers. Based on the data, stress can contribute to the vulnerability of becoming an addict because it activates the HPA axis and increase the dopamine released, which may cause the drug to feel better to a person and make them want to take more of it. There may be a synergy between stress and drugs that changes brain function so that addictive drugs feel more addictive.
You probably noticed I used the word “may” many times and this is because our proposed mechanism requires a lot more testing. In fact, we barely even scratched the surface with this discussion! There are literally hundreds more papers looking at many other details just on stress and addiction. Hopefully this post and the previous two can give you a little appreciation for the difficultly in learning anything about how addiction really works and what specific changes occur in the brain from drug use! Science is a challenging and time-consuming pursuit but also totally worth it!
To wrap up our discussion on stress and addiction, I’ll address some questions/criticisms that you might have with the research papers in this and previous two posts.
Q & A
Some questions about the research you might have and my answers:
Q: Only the psychostimulants cocaine and amphetamine were looked at in these papers. Does stress have the same effects on other drugs of abuse?
A: Yes. The effect of stress is the same with nearly all drugs of abuse tested including the opioid morphine and heroin, alcohol, and nicotine. The neural machinery that is responsible for enhancing the addictive powers of drugs is common to all drugs of abuse.
Q: Only the initial stages of drug taking were looked at in these papers. That is to say, the role of stress was only discussed in the initiation of addiction. How does this translate into progression to full blown addiction?
A: The effect of stress is consistent regardless of where you are on the addiction continuum: stress enhances the reinforcing properties of drugs of abuse. That is to say, stress makes the pleasurable feeling from drugs more pleasurable. However, in humans, you will never get as clear of an effect (that means, easily testable) as you will in laboratory animals. Humans experience many different types of stress throughout a single day and the specific effect of stress on drug taking depends on the type/length/frequency of the stress and other environmental factors. Nevertheless, in controlled clinical studies, changes in HPA axis function as a result of drug use have been widely reported. A feed-forward mechanism exists in which stress promotes drug taking and then drug effects the stress response so that the next stressor has a greater effect on drug taking, etc.
Q: Can stress trigger relapse?
A: Yes, this is one of the most well studied effects of stress on drug taking: stress can trigger drug cravings in abstinent individuals. In the laboratory, an animal can be taught to lose its self-administration behavior by switching the drug to a neutral substance like saline. Therefore, when the animal nose pokes it does not get drug and eventually it doesn’t nose poke at all. This is called extinction. Amazingly, if you stress an animal with foot-shocks or some other phase and then test it’s self-administration behavior the animal will go back to lever pressing again!
Thanks again for reading! If you stuck through all three of Stress and Addiction posts please comment or email me. I would love to know!
Next time: Doping on Dopamine.











